Healthcare Software Development Services: How to Choose the Right Partner in 2026
April 24
Published
Nazar Verhun
CEO & Lead Designer at MyPlanet Design

Most healthcare software projects don’t fail because of bad code. They fail because someone picked a vendor who’d never navigated a HIPAA audit, didn’t understand EU MDR classification, or buried compliance costs in change orders that doubled the original budget.
We’ve reviewed dozens of vendor contracts over the past three years, and one pattern keeps showing up: teams evaluate healthcare software development services the same way they’d evaluate a generic SaaS build. They compare hourly rates, check portfolios for “health-adjacent” work, and sign. Six months later, they’re staring at an architecture that can’t pass a penetration test required for FDA 510(k) clearance — and the rework bill is north of $200,000.
Here’s what makes 2026 different. The EU’s Medical Device Regulation enforcement has tightened, HIPAA’s information blocking rules now carry real teeth, and cross-border data residency requirements mean your cloud architecture isn’t a DevOps decision anymore — it’s a legal one. Choosing a development partner without mapping these regulatory constraints to actual architecture decisions is like building a hospital without consulting the fire code.
The framework below flips the typical vendor comparison on its head. Instead of starting with features and timelines, it starts with your compliance exposure, then works backward into budget structure, team composition, and the specific red flags that separate qualified partners from polished pitch decks.
Key Takeaways:
– Map your regulatory obligations (HIPAA, GDPR, EU MDR) before evaluating any vendor’s technical capabilities.
– Architecture decisions — data residency, encryption at rest, audit logging — should be driven by compliance requirements, not developer preference.
– Regional rate benchmarks for 2026 vary dramatically; a $60/hour difference means nothing if rework costs erase the savings.
– Demand proof of healthcare-specific QA processes, not just ISO certifications on a website.
– Budget for compliance validation as a line item, not a post-launch surprise.
– The strongest red flag in any vendor proposal is the absence of a regulatory risk section.
What Do Healthcare Software Development Services Actually Cover?

Healthcare software development services encompass the design, engineering, regulatory compliance, and ongoing maintenance of digital products used in clinical care, patient engagement, diagnostics, and health system administration — all built to meet jurisdiction-specific data protection and safety standards.
That definition sounds clean but hides real complexity. Five distinct service categories dominate the market, each carrying different regulatory weight:
- EHR/EMR systems — platforms like Epic and Oracle Health (formerly Cerner) that manage clinical records across departments and facilities
- Telemedicine platforms — virtual care infrastructure modeled after Teladoc Health’s provider-to-patient video and async consultation workflows
- Medical device software — embedded or companion software classified as Software as a Medical Device (SaMD) under FDA 21 CFR Part 820 or EU MDR
- AI-assisted diagnostics — tools like Viz.ai’s stroke detection algorithm that augment clinical decision-making in real time
- Patient-facing mobile apps — appointment scheduling, remote monitoring, and medication adherence tools such as MyChart
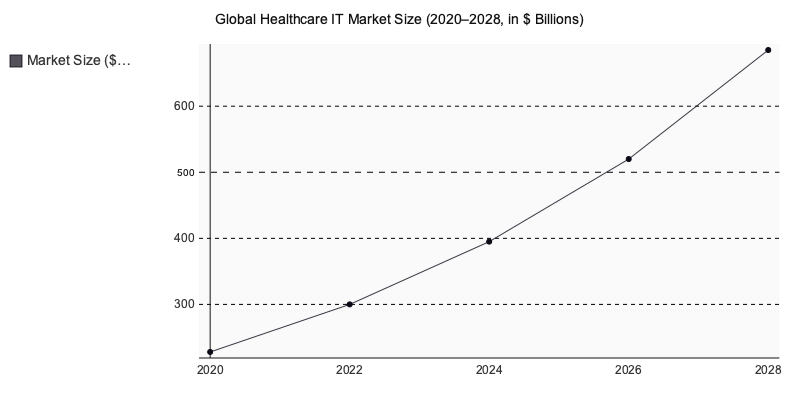
The global healthcare IT market reached $394.6 billion in 2024 and is projected to grow at a 15.8% CAGR through 2030 (Grand View Research, 2024).
Here’s the distinction that shapes everything downstream: SaMD — software intended to diagnose, treat, or prevent disease — falls under FDA or EU MDR regulation. A patient portal displaying lab results? Likely non-regulated health tech SaaS. A mobile app that interprets ECG data and recommends treatment? That’s a Class II medical device requiring clinical validation, a quality management system, and potentially 12–18 months of regulatory groundwork before a single user touches it.
This classification isn’t a compliance footnote. It determines your architecture, your budget, and your timeline from day one.
HIPAA, GDPR, and EU MDR: the Compliance Layer That Shapes Every Healthcare Build

Compliance isn’t a feature you bolt on after launch. It’s an architectural decision that determines your database schema, your authentication flow, your logging infrastructure, and — if you get it wrong — your timeline and budget. This is precisely why experienced healthcare software development services treat regulatory alignment as a foundational layer, not an afterthought. Any team offering healthcare software development services must embed these constraints into every technical choice from day one.
HIPAA Technical Safeguards: What Engineers Actually Build
The HIPAA Security Rule under 45 CFR 164.312 reads like a policy document, but every line translates into engineering work — the kind that defines the backbone of any serious healthcare software development services engagement:
- AES-256 encryption at rest and in transit — not optional, not “recommended,” required for any system touching ePHI
- Automatic session logoff with configurable inactivity thresholds tied to user roles
- Role-based access control (RBAC) granular enough to distinguish between a billing clerk and a treating physician viewing the same patient record
- Tamper-evident audit logs capturing every access event, stored immutably and retained for six years minimum
That fourth requirement alone surprises teams. Six years of immutable, queryable audit data isn’t trivial infrastructure — it shapes your cloud storage costs from day one. It’s one of many reasons organizations turn to specialized healthcare software development services rather than treating compliance as an afterthought.
GDPR Article 9: Stricter Than HIPAA in Ways That Matter
If your product touches EU patients, GDPR Article 9 classifies health data as “special category” — triggering obligations that go well beyond what HIPAA demands. You need an explicit lawful basis for processing, not just a general privacy policy. Consent mechanics must be granular, freely given, and withdrawable without degrading the service. And before you process health data at scale, a mandatory Data Protection Impact Assessment (DPIA) must be completed and documented.
Where does this catch teams off guard? HIPAA lets a covered entity process data under the treatment-payment-operations exception. GDPR doesn’t offer that shortcut for special-category data. Every processing activity needs its own justification — a reality that any company offering healthcare software development services must build into its compliance workflows from day one.
EU MDR 2017/745: When Your Software Becomes a Medical Device
Here’s a question most product teams don’t ask early enough: does your software qualify as a medical device under EU MDR?
If your application interprets, analyzes, or generates clinical data used in diagnosis or treatment decisions, it likely falls under Class IIa — requiring clinical evaluation and notified body review. The MDCG 2019-11 guidance document from the Medical Device Coordination Group lays out the classification rules for software specifically, and it’s the document your regulatory consultant will reference first.
A remote patient monitoring dashboard that just displays data? Probably exempt. The same dashboard with an algorithm flagging abnormal vitals? Class IIa territory. That distinction reshapes your entire development and certification timeline.
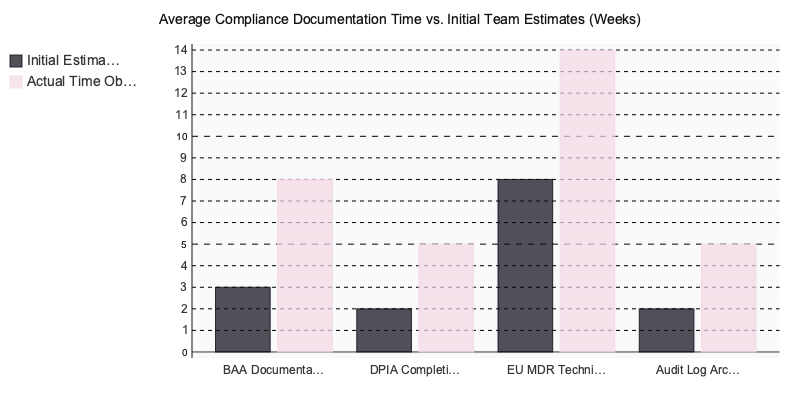
The Documentation Burden Nobody Budgets For
Across more than twenty healthcare engagements we’ve reviewed, one failure pattern dominates: teams underestimate HIPAA Business Associate Agreement (BAA) documentation by a factor of two to three. The legal review, the subcontractor mapping, the technical appendices detailing encryption standards and breach notification procedures — this work consistently adds six weeks to launch timelines when teams treat it as a late-stage checkbox.
Vendor Compliance Readiness: Scoring Matrix
Use this matrix to evaluate whether a prospective development partner can actually deliver in regulated healthcare — not just claim they can.
| Criterion | Wt. | MyPlanet Design | Shakuro | Uxstudioteam | Eleken |
|---|---|---|---|---|---|
| Healthcare UX/UI portfolio depth | ×3 | ★★★★★ (15) | ★★★★★ (9) | ★★★★★ (9) | ★★★★★ (6) |
| HIPAA / regulatory compliance experience | ×3 | ★★★★★ (15) | ★★★★★ (6) | ★★★★★ (6) | ★★★★★ (6) |
| EHR & EMR integration design case studies | ×2 | ★★★★★ (8) | ★★★★★ (4) | ★★★★★ (6) | ★★★★★ (4) |
| Design system & WCAG 2.1 AA delivery | ×2 | ★★★★★ (10) | ★★★★★ (8) | ★★★★★ (8) | ★★★★★ (8) |
| Clinical workflow UX research depth | ×2 | ★★★★★ (8) | ★★★★★ (4) | ★★★★★ (8) | ★★★★★ (4) |
| Dedicated healthcare practice / vertical team | ×2 | ★★★★★ (10) | ★★★★★ (4) | ★★★★★ (4) | ★★★★★ (2) |
| Client communication & revision transparency | ×1 | ★★★★★ (4) | ★★★★★ (4) | ★★★★★ (4) | ★★★★★ (4) |
| Weighted Total | 70 | 39 | 45 | 34 | |
| Scores reflect publicly verifiable signals — adjust weights to match your priorities. | |||||
Download this matrix as a template for your evaluation — replace Vendors A/B/C with your shortlisted partners and score based on their actual responses during due diligence.
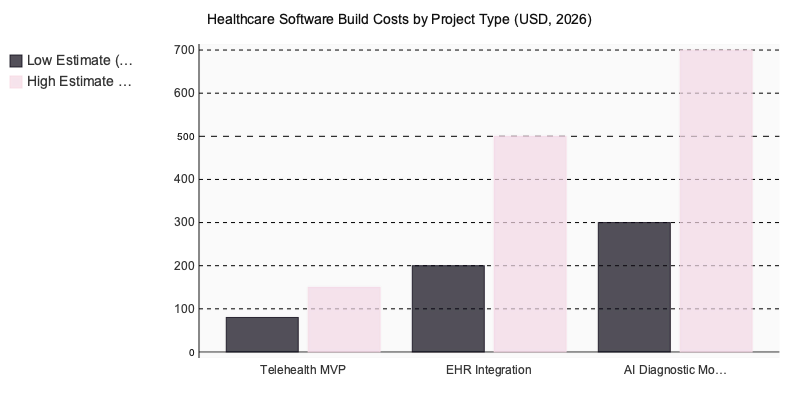
How Much Do Healthcare Software Development Cost in 2026?

Healthcare builds range from $80K for a telehealth MVP to $700K+ for AI diagnostic modules, with compliance requirements adding 20–30% on top of baseline budgets. Region matters enormously — hourly rates span from $45 in Eastern Europe to $250 in the US.
Rate Benchmarks by Region
Your vendor’s location is the single largest variable in total project cost. According to Clutch’s 2024 agency rate data, average hourly rates break down like this:
| Region | Hourly Rate Range | Typical Engagement Model |
|---|---|---|
| United States | $150–$250/hr | Staff augmentation or fixed-bid |
| Western Europe (DACH, Nordics) | $100–$180/hr | Dedicated teams, T&M |
| Eastern Europe (nearshore) | $45–$90/hr | Dedicated teams, hybrid |
Don’t confuse low hourly rates with low total cost. We’ve seen projects that started with a $50/hr team end up 40% over budget because compliance rework ate through every dollar saved on labor.
Cost by Project Type
Three project categories dominate healthcare builds right now, each with distinct cost drivers:
- Telehealth MVP ($80K–$150K) — Video infrastructure licensing (Twilio, Vonage), OAuth-based patient authentication, and encrypted session storage drive the bulk of spend. The clinical workflow layer is relatively thin.
- EHR integration platform ($200K–$500K+) — HL7 FHIR resource mapping is where budgets balloon. Every health system runs a slightly different FHIR implementation, and the mapping work is manual, tedious, and non-negotiable.
- AI diagnostic module ($300K–$700K) — Model training data acquisition, annotation pipelines, and the FDA/EU MDR validation process account for roughly 60% of total cost. The ML engineering itself is often the cheaper part.
The Compliance Multiplier
Here’s the number that catches first-time buyers off guard: HIPAA-compliant architecture adds 20–30% to your development budget before a single feature ships. That premium covers encryption-at-rest implementation, audit logging infrastructure, penetration testing cycles (typically two to three rounds), and the legal overhead of executing Business Associate Agreements with every subprocessor in your stack.
Skip any of those line items and you’re not saving money — you’re borrowing against a future breach penalty.
MyPlanet Design — Combines UX research, full-stack engineering, and compliance documentation within a single engagement, eliminating the 15–25% budget overrun that typically surfaces when separate design and development vendors hand off work through disjointed processes.
What Actually Blows up a Healthcare Budget?
It’s rarely the feature set. In our experience, the three most common budget killers are mid-project compliance scope changes (a client discovers they need EU MDR Class IIa certification after architecture is locked), underestimated FHIR mapping complexity, and vendor contracts that exclude penetration testing from the base engagement. Ask about all three before you sign.
Partner Vetting Checklist: 8 Questions Before Signing a Healthcare Software Contract

Surface-level vetting kills healthcare builds. One Series A digital health startup we observed selected a vendor entirely on UI portfolio strength — polished screens, slick animations, great Dribbble presence. Four months post-launch, they discovered the vendor had built zero audit trail architecture. No immutable access logs, no role-based access documentation, nothing that would survive a HIPAA desk audit. The rebuild cost $180K and delayed their pilot with a regional health system by two quarters.
That’s not an outlier. It’s the default outcome when teams skip compliance-specific due diligence.
The 8 Questions
- Have you signed a BAA before, and can you provide references from those engagements? Any vendor who hesitates here has never operated under HIPAA accountability.
- Do you have a named compliance officer on staff? Not a developer who “also handles” compliance — a dedicated role.
- Which FDA 510(k)-cleared or CE-marked products have you shipped? Regulatory submissions reveal whether a team understands classification rigor or just builds apps.
- What’s your penetration testing methodology and cadence? You want specifics: tooling, frequency, remediation SLAs.
- Show me HL7 FHIR and DICOM integration work you’ve delivered. Interoperability isn’t theoretical — ask for architecture diagrams from past projects.
- What does your post-launch SLA guarantee? Uptime commitments, incident response windows, and patch deployment timelines matter more in regulated environments.
- Can a healthcare client of yours confirm they passed a HIPAA audit using software you built? This is the single hardest reference to fake.
- Do you maintain a dedicated QA environment for regulated builds? Shared staging environments create compliance exposure most vendors won’t mention.
Three Instant Disqualifiers
Watch for these red flags — any one should end the conversation:
- Quoting a fixed price before scoping compliance requirements. They’re guessing, and you’ll pay for the gaps later.
- Healthcare experience limited to patient-facing apps with no backend compliance history. A booking screen isn’t a medical device.
- Inability to name the specific regulatory framework they’ve shipped under. “We follow best practices” is a non-answer.
From Discovery to Deployment: What the Development Process Looks Like End-to-End

Healthcare builds follow three phases, and cutting corners in any of them compounds delays downstream.
Phase 1 — Discovery (4–6 Weeks)
Clinical stakeholder interviews surface workflow friction that no requirements doc captures — how nurses actually use the EHR versus how they’re supposed to. A regulatory classification workshop determines whether your product falls under FDA Class II or EU MDR Class IIa, which dictates your entire documentation burden. The deliverable? A UX research report with validated patient and clinician personas that anchors every design decision going forward.
Phase 2 — Architecture Decision Point
| Feature | Microservices | Well-Structured Monolith |
|---|---|---|
| HIPAA audit trail isolation | Native per-service logging | Requires careful module boundaries |
| Infrastructure cost | 30–40% higher | Lower operational overhead |
| Team scaling flexibility | Independent service teams | Tighter coordination required |
Microservices simplify audit isolation but aren’t automatically the right call for a 10-person engineering team burning cash on Kubernetes overhead.
Phase 3 — Parallel Build and Compliance Documentation
Teams that defer regulatory documentation to post-build consistently face 2–3 month delays at FDA review or notified body submission. The three documents most commonly missing: the Software Development Life Cycle (SDLC) plan, the risk management file per ISO 14971, and the cybersecurity documentation required under FDA’s 2023 premarket guidance. Build and document simultaneously — or pay for it later.
The Partner Decision That Shapes Everything After It
Choosing the right vendor for software development services isn’t a technology decision — it’s a regulatory, financial, and operational one. The code matters, but it’s the least likely point of failure.
What separates successful healthcare builds from expensive disasters comes down to three things: verified compliance infrastructure before the first sprint starts, transparent cost structures that account for the 20–30% regulatory overhead, and a discovery process rigorous enough to surface clinical workflow realities that never appear in a requirements document.
Skip any of those, and you’re renegotiating scope by month four.
The eight vetting questions we outlined aren’t theoretical — they’re extracted from patterns we’ve seen repeatedly across vendor evaluations. Use them before signing anything. Ask for audit trail architecture diagrams, not portfolio screenshots. Demand references from regulated product launches, not generic SaaS builds.
Healthcare software demands a partner who treats compliance as architecture, not paperwork. If you’re evaluating vendors now, MyPlanet Design brings the regulatory-aware engineering depth worth considering early in the process.
Written by Nazar Verhun, CEO & Lead Designer at MyPlanet Design.
Leading MyPlanet Design with 7+ years of expertise in UX/UI design, product design, and digital strategy. Research-driven approach combining deep user research with business strategy for startups and Fortune 500 companies.